|
1/23/2024 0 Comments Carbon 14 beta decay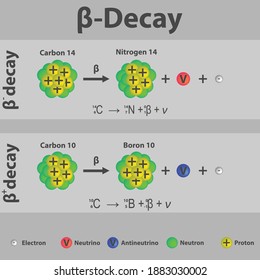
The electron, #e#, carries off a negative charge leaving the nucleus with an additional positively charged proton and one fewer neutron. ISBN: 978-2759800414.See, which states that carbon-14 decays thusly: Robert Reed Burn, Introduction to Nuclear Reactor Operation, 1988.Nuclear Reactor Engineering: Reactor Systems Engineering, Springer 4th edition, 1994, ISBN: 978-0412985317 Stacey, Nuclear Reactor Physics, John Wiley & Sons, 2001, ISBN: 0- 471-39127-1. Baratta, Introduction to Nuclear Engineering, 3d ed., Prentice-Hall, 2001, ISBN: 8-1. Lamarsh, Introduction to Nuclear Reactor Theory, 2nd ed., Addison-Wesley, Reading, MA (1983). DOE Fundamentals Handbook, Volume 1 and 2. Department of Energy, Nuclear Physics and Reactor Theory. Martin, James E., Physics for Radiation Protection 3rd Edition, Wiley-VCH, 4/2013.Stabin, Michael G., Radiation Protection and Dosimetry: An Introduction to Health Physics, Springer, 10/2010.Knoll, Glenn F., Radiation Detection and Measurement 4th Edition, Wiley, 8/2010.When the biological system dies, it stops exchanging carbon with its environment, and from that point onwards the amount of carbon-14 it contains begins to decrease as the carbon-14 undergoes radioactive decay. The annual dose from carbon-14 is estimated to be about 12 μSv/year.Īs long as the biological system is alive the level is constant due to constant intake of all isotopes of carbon. The decay of a carbon-14 atom inside DNA in one person happens about 50 times per second, changing a carbon atom to one of nitrogen. However, a carbon atom is in the genetic information of about half the cells, while potassium is not a component of DNA. Note that, biological half-life is the time taken for the amount of a particular element in the body to decrease to half of its initial value due to elimination by biological processes alone. Carbon-14 is present in the human body (13kg of carbon in 70kg human) at a level of about 3700 Bq (0.1 μCi) with a biological half-life of 40 days. 
As a result, all living biological substances contain the same amount of C-14 per gram of carbon, that is 0.3 Bq of carbon-14 activity per gram of carbon. On average just one out of every 1.3 x 10 12 carbon atoms in the atmosphere is a radioactive carbon-14 atom. As a result, carbon-14 is continuously formed in the upper atmosphere by the interaction of cosmic rays with atmospheric nitrogen. These neutrons produced in these collisions can be absorbed by nitrogen-14 to produce an isotope of carbon-14:Ĭarbon-14 can also be produced in the atmosphere by other neutron reactions, including in particular 13C(n,γ)14C and 17O(n,α)14C. These cosmic rays interact with nuclei in the atmosphere, and produce also high-energy neutrons. Besides, the atmosphere is constantly bombarded with high energy cosmic rays, consisting of protons, heavier nuclei, or gamma rays. Our atmosphere contains many gases, including nitrogen-14. Its presence can be explained by the following simple observation. In spite of this short half-life compared to the age of the earth, carbon-14 is a naturally occurring isotope. Radioactive carbon-14 has a half-life of 5730 years and undergoes β− decay, where the neutron is converted into a proton, an electron, and an electron antineutrino: Beta decay of C-14 nucleus. The only cosmogenic radionuclide to make a significant contribution to internal exposure of human is carbon-14.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
